| [1]李素萍.骨质疏松动物模型的研究现状[J].中国组织工程研究与临床康复,2011,15(20):3767-3770.
[2]Maeda SS, Lazaretti-Castro M. An overview on the treatment of postmenopausal osteoporosis. Arq Bras Endocrinol Metabol. 2014;58(2):162-171.
[3]王东,顾建红,刘宗平.组织蛋白酶K在骨吸收中的作用研究进展[J].动物医学进展,2014,35(6):114-116.
[4]Costa AG, Cusano NE, Silva BC, et al. Cathepsin K: its skeletal actions and role as a therapeutic target in osteoporosis. Nat Rev Rheumatol. 2011;7(8):447-456.
[5]Kwak HB, Kim JY, Kim KJ, et al. Risedronate directly inhibits osteoclast differentiation and inflammatory bone loss. Biol Pharm Bull. 2009;32(7):1193-1198.
[6]张鹏,刘强,董伟,等.唑来膦酸对成骨细胞单核巨噬细胞共培养体系中Atp6vod2基因表达及破骨细胞生成的抑制作用[J].实用口腔医学杂志,2013,29(6):766-769.
[7]陈国仙,王国荣,林宗锦,等.核因子κB受体活化因子配体诱导形成的成熟破骨细胞[J].中国组织工程研究,2013,17(24): 4380- 4385.
[8]刘强,董伟,戚孟春,等.阿仑膦酸盐对破骨细胞生成的抑制及钙离子激动剂的拮抗效应[J].实用口腔医学杂志,2012;2:182-186.
[9]Pang M, Martinez AF, Fernandez I, et al. AP-1 stimulates the cathepsin K promoter in RAW 264.7 cells. Gene. 2004(1-2): 151-158.
[10]李鹏,林珏杉,张鹏,等.唑来膦酸对破骨细胞分化中CAM、CAMKⅡ基因表达的抑制作用[J].中华口腔医学杂志,2013,48(11): 694-698.
[11]王翔,郭海玲,赵咏芳,等.阿仑膦酸钠改善去卵巢大鼠的骨基质结构[J].中国组织工程研究,2013;17(50):8621-8628.
[12]褚建国,王季军.骨质疏松骨代谢的OPG/RANKL/RANK作用机制[J].中国老年学杂志,2013,33(20):5015-5017.
[13]Arai M, Nagasawa T, Koshihara Y, et al. Effects of beta-adrenergic agonists on bone-resorbing activity in human osteoclast-like cells. Biochem Biophys Acta. 2003;1640: 137-142.
[14]Leung P, Pickarski M, Zhuo Y, et al. The effects of the cathepsin K inhibitor odanacatib on osteoclastic bone resorption and vesicular trafficking. Bone. 2011;49(4):623-635.
[15]Troen BR. The regulation of cathepsin K gene expression. Ann N Y Acad Sci. 2006,1068:165-172.
[16]Li Z, Hou WS, Escalante-Torres CR, et al. Collagenase activity of cathepsin k depends on complex fromation with chordroitin sulfate. J Biol Chem. 2002;277:28669-28876.
[17]何伟涛,刘康,孙金谮,等.组织蛋白酶K与骨质疏松症治疗的研究进展[J].中国骨质疏松杂志,2008,14(9):670-673.
[18]Wilson SR,Peters C,Saftig P, et al.Cathepsin K Activity-dependent Regulation of Osteoclast Actin Ring Formation and Bone Resorption. J Biol Chem. 2009;284(4): 2584-2592.
[19]Fumiyo I, Riko N, Takuma M, et al. Critical roles of c-Jun signaling in regulation of NFAT family and RANKL-regulated osteoclast differentiation. J Clin Invest. 2004;114(4):475-484.
[20]Balkan W, Martinez AF, Fernandez I, et al. Identification of NFAT binding sites that mediate stimulation of cathepsin K promoter activity by RANK ligand. Gene. 2009;446(2):90-98.
[21]Huang H, Ryu J, Ha J, et al. Osteoclast differentiation requires TAK1 and MKK6 for NFATc1 induction and NF-kappaB transactivation by RANKL. Cell Death Differ. 2006; 13:1879-1891.
[22]Armstrong AP, Tometsko ME, Glaccum M, et al. A RANK/TRAF6-dependent signal transduction pathway is essential for osteoclast cytoskeletal organization and resorptive function. J Biol Chem. 2002;277(46):44347-44356.
[23]Komarova SV, Pilkington MF, Weidema AF, et al. RANK ligand-induced elevation of cytosolic Ca2+ accelerates nuclear translocation of NF-kB inosteoclasts. J Biol Chem. 2003;278: 8286-8293.
[24]董强,夏茜,周建奖,等.流体切应力作用下破骨细胞组织蛋白酶K的表达[J].实用口腔医学杂志,2013,29(4):459-462.
[25]张大威,程岩,张金超,等.淫羊藿苷对破骨细胞的分化及骨吸收功能的影响[J].中国药理学通报,2007,23(4):463-467.
[26]孙彦,李兴朱,亦垄.葡萄糖对大鼠骨髓破骨细胞骨吸收功能的影响[J].中华内分泌代谢杂志,2009,25(5):549-551.
[27]Choi J, Choi SY, Lee SY, et al. Caffeine enhances osteoclast differentiation and maturation through p38 MAP kinase/Mitf and DC-STAMP/CtsK and TRAP pathway. Cell Signal. 2013; 25(5):1222-1227.
[28]曲新华,郝永强.双膦酸盐在骨科相关疾病治疗中的地位及再认识[J].中华骨科杂志,2014,34(1):78-80.
[29]胡春明,王成学,莫丽娟,等.骨质疏松药物治疗进展[J].中国老年学杂志,2008,28(21):2178-2180.
[30]Whitfield JF, Mormey PS. Small bone-building fragments of parathyroid homone:New therapeutic agents for osteoporosis. Trend Pharmacol Sci.1995;16:382-395.
[31]Svanström H, Pasternak B, Hviid A. Use of strontium ranelate and risk of acute coronary syndrome: cohort study. Ann Rheum Dis. 2014;73(6):1037-1043.
[32]Leder BZ, Tsai JN, Uihlein AV, et al. Two years of Denosumab and teriparatide administration in postmenopausal women with osteoporosis (The DATA Extension Study): a randomized controlled trial. J Clin Endocrinol Metab. 2014;99(5):1694-1700.
[33]Dalbeth N, Aati O, Gamble GD, et al. Zoledronate for prevention of bone erosion in tophaceous gout: a randomised, double-blind, placebo-controlled trial. Ann Rheum Dis. 2014; 73(6):1044-1051.
[34]McClung MR, Grauer A, Boonen S, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014;370(5):412-420.
[35]Tüzün ?, Akyüz G, Eskiyurt N, et al. Impact of the training on the compliance and persistence of weekly bisphosphonate treatment in postmenopausal osteoporosis: a randomized controlled study. Int J Med Sci. 2013;10(13):1880-1887.
[36]Mok JO, Jung CH, Kim CH, et al. Endoscopic comparison of alendronate alone and the enteric-coated alendronate with calcitriol combination in postmenopausal Korean females. Korean J Intern Med. 2013;28(6):694-700.
[37]Castro-Lionard K, Dargent-Molina P, Fermanian C, et al. Use of calcium supplements, vitamin D supplements and specific osteoporosis drugs among French women aged 75-85 years: patterns of use and associated factors. Drugs Aging. 2013; 30(12):1029-1038.
[38]Kachnic LA, Pugh SL, Tai P, et al. RTOG 0518: randomized phase III trial to evaluate zoledronic acid for prevention of osteoporosis and associated fractures in prostate cancer patients. Prostate Cancer Prostatic Dis. 2013;16(4):382-386.
[39]Bonnick S, De Villiers T, Odio A, et al. Effects of odanacatib on BMD and safety in the treatment of osteoporosis in postmenopausal women previously treated with alendronate: a randomized placebo-controlled trial. J Clin Endocrinol Metab. 2013;98(12):4727-4735.
[40]Ooi HL, Briody J, Biggin A, et al. Intravenous zoledronic Acid given every 6 months in childhood osteoporosis. Horm Res Paediatr. 2013;80(3):179-184.
[41]Sjöberg C, Wallerstedt SM. Effects of medication reviews performed by a physician on treatment with fracture-preventing and fall-risk-increasing drugs in older adults with hip fracture-a randomized controlled study. J Am Geriatr Soc. 2013;61(9):1464-1472.
[42]Lazzari AA, Dussault PM, Thakore-James M, et al. Prevention of bone loss and vertebral fractures in patients with chronic epilepsy--antiepileptic drug and osteoporosis prevention trial. Epilepsia. 2013;54(11):1997-2004.
[43]Sato Y, Honda Y, Iwamoto J, et al. Comparison of non-vertebral fracture between minodronate and risedronate therapy in elderly female patients with Alzheimer disease. J Musculoskelet Neuronal Interact. 2013;13(3):346-352.
[44]Gatti D, Rossini M, Viapiana O, et al. Teriparatide treatment in adult patients with osteogenesis imperfecta type I. Calcif Tissue Int. 2013;93(5):448-452.
[45]Polidoro S, Broccoletti R, Campanella G, et al. Effects of bisphosphonate treatment on DNA methylation in osteonecrosis of the jaw. Mutat Res. 2013;757(2):104-113.
[46]Wan X, Zhao Y, Burge R, et al. Actions of osteoporosis treatments on bone histomorphometric remodeling: a two-fold principal component analysis. Osteoporos Int. 2013;24(12): 3011-3019.
[47]Lang JM, Wallace M, Becker JT, et al. Arandomized phase II trial evaluating different schedules of zoledronic acid on bone mineral density in patients with prostate cancer beginning androgen deprivation therapy. lin Genitourin Cancer. 2013; 11(4):407-445.
[48]Recknor C, Czerwinski E, Bone HG, et al. Denosumab compared with ibandronate in postmenopausal women previously treated with bisphosphonate therapy: a randomized open-label trial. Obstet Gynecol. 2013;121(6): 1291-1299.
[49]Hari P, DeFor TE, Vesole DH, et al. Intermittent zoledronic Acid prevents bone loss in adults after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2013;19(9):1361-1367.
[50]Chung HY, Koo J, Kwon SK, et al. Early changes in 25-hydroxyvitamin D levels and bone markers after monthly risedronate with cholecalciferol in Korean patients with osteoporosis. Clin Interv Aging. 2013;8:597-603. |
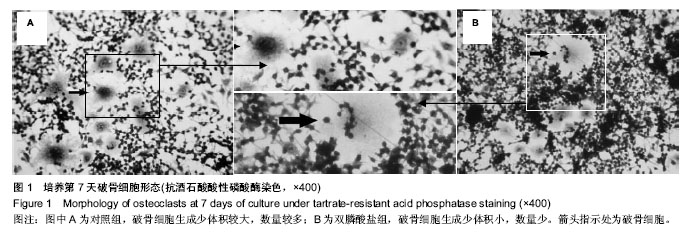
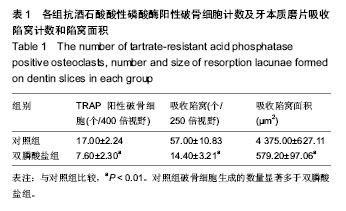
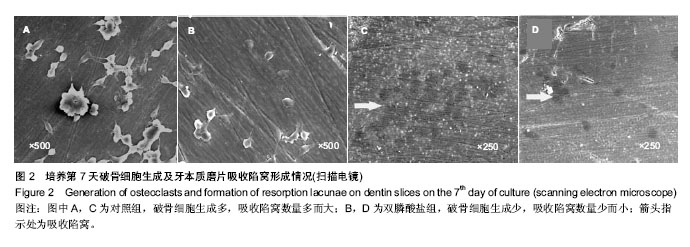
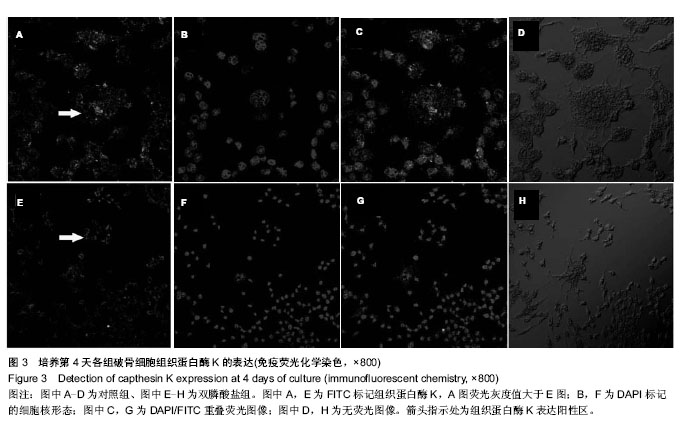

.jpg)